Zero-Defect Label Production: Automated Print Quality Inspection Machine
Description
Label Quality Inspection Machine
Zero-Defect Production Starts Here
AI-powered automated vision systems for inline and offline label defect detection — engineered for pharmaceutical, food, and cosmetic label converters.
Industry Problem

Why Manual Label Inspection Is No Longer Enough
As label complexity increases and tolerance windows shrink, human vision inspection creates unacceptable production risk.
In the pharmaceutical and food industries, a single mislabeled shipment can trigger a full product recall — costing manufacturers an average of $10M+ per incident. Fonts as small as 5.5pt and micro-barcodes require resolution and consistency that no human inspector can sustain at production speeds.
Traditional label production relied on end-of-line spot checks by trained human inspectors — effective for low-volume, low-complexity jobs. But the modern label market has fundamentally changed: shorter runs, more SKUs, tighter regulatory requirements, and digital printing technologies that demand 100% inspection at machine speeds.
The shift from reactive to proactive quality control is now a competitive necessity. Converters who deploy automated label inspection report a 60–85% reduction in customer complaints, near-elimination of recall risk, and measurable gains in press uptime by catching systemic defects before they consume an entire roll.
Machine Demonstration Video
Defect Detection

What Defects Can This Machine Detect?
Comprehensive machine vision coverage across all common flexographic and digital label printing failure modes.
Missing Text / Print
Detects absent or incomplete text fields, critical for pharma lot numbers and expiry dates.
Color Deviation (ΔE)
Spectrophotometric comparison flags ΔE >1.5 against master standard, per brand color specs.
Barcode / QR Verification
ISO/IEC 15415 grade verification confirms scanability. Detects damage, distortion, and quiet zone violations.
Registration Misalignment
Sub-0.1mm overprint registration check for multi-color labels on narrow-web flexo presses.
Ink Smudge & Voids
High-contrast LED illumination reveals ink smears, hickeys, voids, and contamination spots.
Foil / Cold Stamp Defects
Dedicated reflective inspection channel detects hot foil voids, delamination, and cold stamp tears.
Bubbles & Scratches
Surface uniformity analysis catches substrate bubbles, laminate lift, and mechanical scratch marks.
Die Cut Accuracy
Label outline conformance check — detects over-cut, under-cut, and rotary die misalignment.
Overflow / Bleed
Detects ink spread beyond print boundaries that compromises branding and regulatory print zones.
Industry-Specific Defect Sensitivity
Pharmaceutical labels: Mandatory 100% barcode verification (GS1-128, DataMatrix), font legibility at 5.5pt minimum, lot/expiry date presence check.
Food & beverage labels: Allergen statement completeness, nutritional table presence, country-of-origin compliance under FDA 21 CFR Part 101.
Cosmetic labels: INCI ingredient list integrity, claim consistency, EU Cosmetics Regulation 1223/2009 compliance fields.
Electronic / industrial labels: UL/CE mark verification, warning symbol integrity, RoHS compliance icon detection.
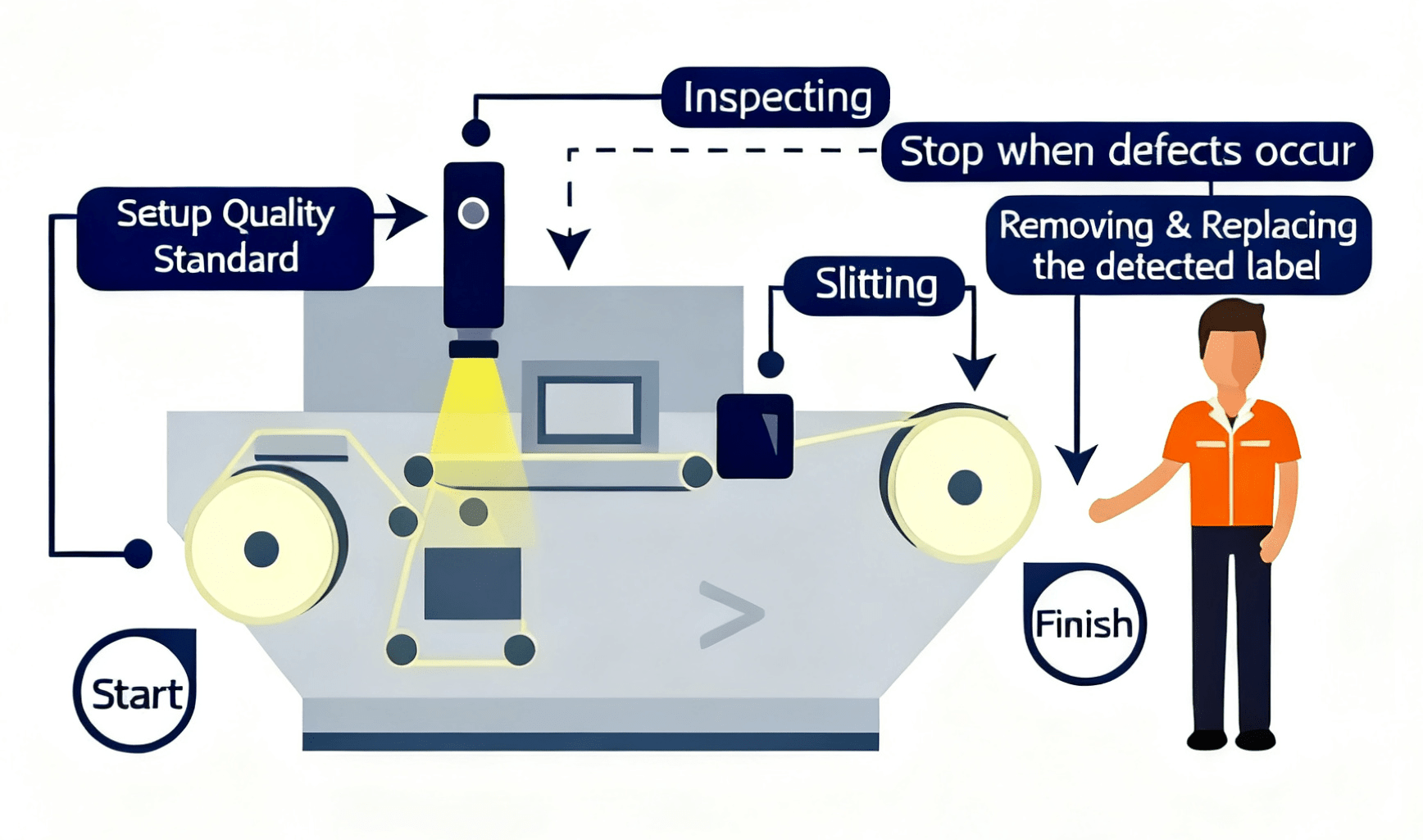
How the Label Inspection System Works
A closed-loop machine vision pipeline from substrate entry to defect-flagged rewind — all in real time.
Unwind & Tension Control
Precision dancer rollers maintain constant web tension for stable imaging.
Multi-Channel Illumination
Stroboscopic LED (white, UV, IR) illuminates each label for maximum defect contrast.
8K Line Scan Imaging
High-resolution CCD camera captures full label width at up to 150 m/min.
AI Vision Processing
Image processing software compares each label to the golden master template in <5ms.
Auto-Stop & Mark
Defect triggers automatic web stop or ink-jet marking for post-process removal.
Data & Report
OPC-UA / MES integration logs defect map, position, classification, and production metrics.
Machine Vision Architecture: What Sets It Apart
Unlike basic 2D camera systems, modern label inspection platforms deploy multi-spectral imaging — combining visible, UV, and near-infrared illumination channels to detect defects that are invisible under a single light source. UV excitation, for instance, reveals invisible UV varnish coverage and security ink features; IR illumination detects density variations beneath surface coatings.
The inspection software uses a hierarchical template matching algorithm rather than simple pixel-by-pixel comparison. This makes the system tolerant of expected press variations (slight ink density fluctuation, substrate stretch) while remaining hypersensitive to real defects. The result is an exceptionally low false-reject rate — typically below 0.01% — which eliminates the economic cost of false alarms that plague lower-grade systems.
Key Features of Our Label Inspection System
Engineered for narrow-web converters demanding zero-defect output with minimal operator intervention.
High-Resolution 8K Line Scan Camera
Industrial-grade CCD sensors with 8192 pixel resolution deliver sub-0.04mm ground sampling distance at full web width — detecting even the finest dot gain defects and hairline scratches invisible to the naked eye.
Real-Time Processing at 150 m/min
Dedicated FPGA image processing hardware delivers <5ms frame analysis latency. Machine stops in under 300ms from defect detection — minimizing waste to typically less than 1.5 label pitches per event.
AI-Assisted Template Learning
Machine learning algorithms build adaptive inspection templates from initial good labels, automatically compensating for substrate expansion, registration drift, and press colour variation — reducing setup time from hours to under 15 minutes.
Integrated Barcode & OCR Verification
ISO-grade 1D/2D barcode decoding (including GS1-128, ITF-14, QR, DataMatrix) plus OCR for alphanumeric strings. Lot number, expiry date, and batch code verification in a single pass.
MES / ERP Integration & OEE Reporting
OPC-UA, RS-232, and Ethernet interfaces connect to plant MES/ERP systems. Automatic SPC charting, defect Pareto reports, and ISO 9001-compliant quality records are generated per production job.
Automatic Defect Marking & Removal
Ink-jet defect flagging, automatic splice-tape application, or programmable stop-and-mark modes allow operator-free defect isolation — compatible with downstream label die-cutters and rewinders.
Technical Specifications
Standard configuration for narrow-web self-adhesive label inspection. Custom widths and speeds available on request.
| Parameter | Specification | Notes |
|---|---|---|
| Web Width | Up to 520 mm (20.5") | Customizable to 650mm |
| Max Inspection Speed | 150 m/min (492 ft/min) | Full resolution maintained |
| Camera Resolution | 8,192 pixels / 8K | CCD line scan sensor |
| Minimum Defect Size | 0.1 mm² (at 100 m/min) | Speed-dependent sensitivity |
| Illumination | Multi-spectral LED (White, UV, IR) | Stroboscopic, zero flicker |
| Color Inspection | ΔE < 1.5 detection threshold | CIELab color space |
| Barcode Standards | ISO/IEC 15415 & 15416 | Grade A–F report |
| OCR / OCV | Yes — alphanumeric, 5.5pt min | Multi-font library |
| Unwind Diameter | Up to 1,000 mm | Core sizes: 3", 6" |
| Rewind Diameter | Up to 1,000 mm | Programmable tension profiles |
| Meter Counter | Automatic — sets length & quantity | ±0.1m accuracy |
| Communication | OPC-UA, RS-232, Ethernet, USB | MES/ERP ready |
| Power Supply | Three-phase 380V / 50Hz | 220V/60Hz optional |
| Machine Footprint | Approx. 2,200 × 900 × 1,600 mm | W×D×H, excluding HMI stand |
Inline vs Offline Label Inspection: Which Do You Need?
Both inspection modes serve distinct roles in a zero-defect quality management strategy.
| Criteria | Inline Inspection | Offline Inspection |
|---|---|---|
| When it runs | During the print run, mounted on press | After printing, on standalone rewinder |
| Defect detection | ✓ Catches defects in real time, stops waste immediately | Detects defects post-production, before shipment |
| Press productivity impact | Minimal — integrated into press stop/start logic | ✓ None — fully decoupled from press |
| Defect reporting depth | Real-time alerts + shift-level summary | ✓ Full batch defect map + ISO quality certificate |
| Best for | Long-run flexo jobs, pharmaceutical mandates | Short-run digital, incoming inspection of bought-in labels |
| Integration complexity | Requires press communication interface | ✓ Plug-and-play standalone operation |
| Recommended for | FDA/GMP regulated labels, pharma, food | General commercial, short-run cosmetic labels |
Best-Practice Recommendation
For pharmaceutical and food converters operating under GMP: deploy inline inspection on the press + offline inspection for final release. This gives both real-time waste control and a signed-off quality record per batch.
For general commercial converters: a standalone offline inspection rewinder with 100% vision coverage provides a high-quality gate before shipment without capital investment in press integration.
For digital label presses (HP Indigo, Epson SurePress, Canon Océ): inline inspection is essential since digital substrates have high per-unit cost — catching a defect early prevents expensive roll waste.
Industries That Rely on Label Quality Inspection
Automated label inspection is mandated or strongly recommended across highly regulated and brand-sensitive industries.
Pharmaceutical & Healthcare
Regulatory bodies including the FDA (21 CFR Part 211), EMA, and ANVISA require 100% label accuracy for prescription drugs, OTC products, and medical devices. Automated inspection systems provide the audit trail required for GMP compliance. Key verification requirements include barcode grade (GS1-128 or DataMatrix for serialization), lot number/expiry date OCR, and tamper-evident label integrity.
Food & Beverage
Allergen mislabeling is the leading cause of food recalls in North America and Europe. Inspection systems verify that mandatory advisory statements (e.g., "Contains: Milk, Wheat, Soy") are present on every label — catching plate-swap and version-control errors before they reach retail shelves.
Cosmetics & Personal Care
EU Cosmetics Regulation 1223/2009 and US FDA cosmetics labeling rules require specific ingredient list formatting, country of origin, and responsible person information. Automated inspection catches layout errors, missing fields, and foreign-language version mix-ups common in multi-market label runs.
Electronics & Industrial
UL recognition marks, CE marking, and RoHS compliance symbols must meet precise dimensional tolerances to remain legally valid. Machine vision inspection verifies symbol integrity, serial number uniqueness (in serialized label production), and ESD warning label placement accuracy.
Frequently Asked Questions
Common questions from label converters evaluating automated inspection systems.
Ready to Achieve Zero-Defect Label Production?
Our applications engineers will recommend the right inspection configuration for your press, substrate, and regulatory requirements — and provide a full ROI analysis.


